 The compilation includes data for the neutral and singly-ionized atoms of all elements hydrogen through einsteinium ( Z 1-99). ^ "Table of Refractive Indices and Double Refraction of Selected Gems - IGS". Many materials have a well-characterized refractive index, but these indexes often depend strongly upon the frequency of light, causing optical dispersion.Standard refractive index measurements are taken at the 'yellow doublet' sodium D line, with a wavelength () of 589 nanometers. Introduction This handbook is designed to provide a selection of the most important and frequently used atomic spectroscopic data in an easily accessible format.In general, an index of refraction is a complex number with both a real and imaginary part, where the latter indicates the strength of absorption loss at a particular wavelength-thus, the imaginary part is sometimes called the extinction coefficient k " (PDF). Record the observed colors and wavelength values of the elements emission. Thus, it is especially important to cite the source for an index measurement if precision is required. Use the spectroscope to observe the color of flame for each of the given elements. The line at 589.0 has twice the intensity of the line at 589.6 nm. From the energy level diagram it can be seen that these lines are emitted in a transition from the 3p to the 3s levels. Wavelength isolation is by use of a simple narrow-bandpass interference filter that is. There are also weaker dependencies on temperature, pressure/ stress, etc., as well on precise material compositions (presence of dopants, etc.) for many materials and typical conditions, however, these variations are at the percent level or less. The sodium spectrum is dominated by the bright doublet known as the Sodium D-lines at 588.9950 and 589.5924 nanometers. generates strong emission only from the most easily excited elements.  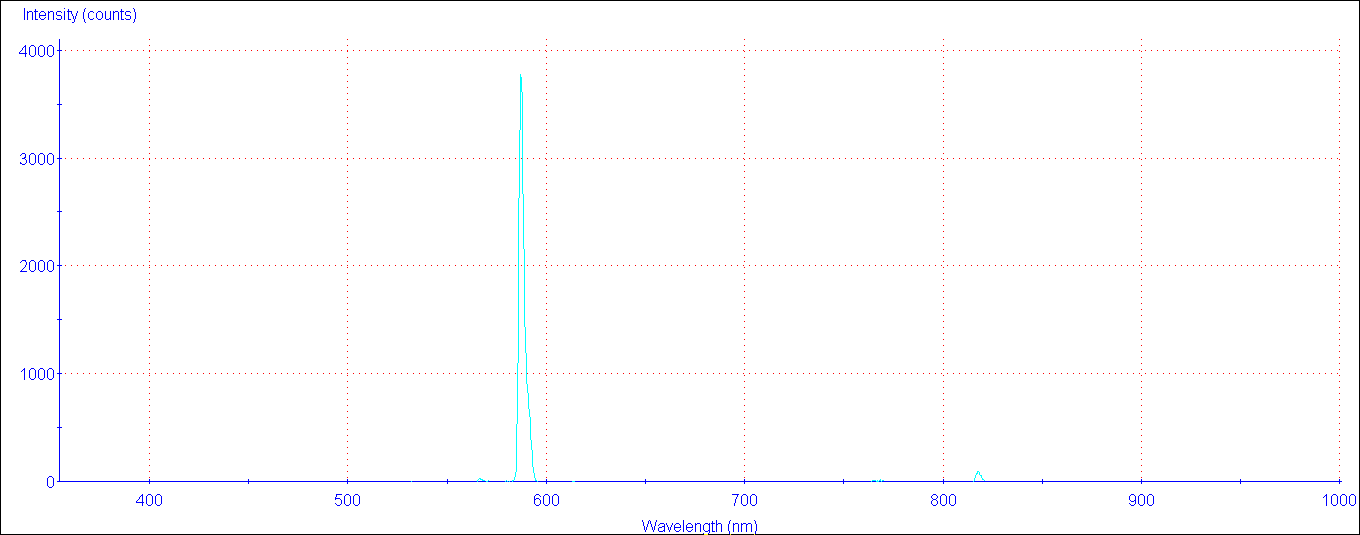
Standard refractive index measurements are taken at the "yellow doublet" sodium D line, with a wavelength (λ) of 589 nanometers. The element sodium, after having been excited electrically, emits an amount of energy equal to 3.37 x 1019 J/atom. The sodium spectrum is dominated by thebright doublet known as the Sodium D-lines at 588.9950 and 589.5924nanometers. 
Many materials have a well-characterized refractive index, but these indexes often depend strongly upon the frequency of light, causing optical dispersion. element like sodium which are in the gaseous state, the gaseous atoms absorb photons from the white light of specific frequency/wavelength (and hence. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
